Picornavirus Polyprotein Processing.A common, if not ubiquitous, strategy of positive strand RNA viruses is to encode some, or all, of their proteins in the form of polyproteins. This strategy is not confined to viruses, as both eu- and prokaryotes also (rarely) use polyproteins in protein biogenesis.
Cellular polyproteins may comprise different proteins i.e proopiomelanocortin (POMC) and adrenocorticotropic hormone (ACTH), or, may contain multiple copies of the same protein i.e. yeast prepro-a-mating factor.
Whilst cellular polyproteins are processed by cellular enzymes post-translationally, the presence of proteinases within positive strand RNA virus polyproteins can bring about both co-translational (in cis) and post-translational (in trans) proteolytic processing.
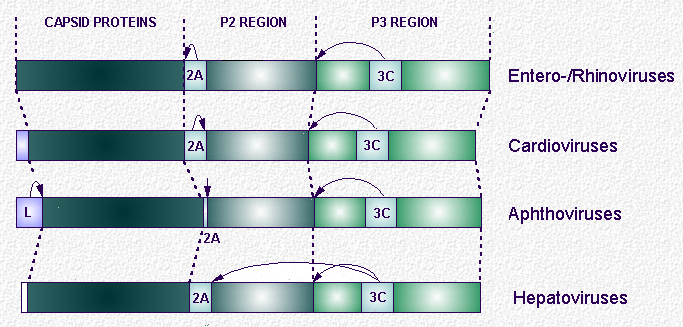
The picornaviruses encode all of their proteins in a single, long, open reading frame. Full-length translation products are not observed, however, due to extremely rapid co-translational, intramolecular (in cis), or ‘primary’ cleavages.
Subsequent ‘secondary’ processing of the primary cleavage products may occur either in cis or in an intermolecular fashion (in trans). Such processing of the replicative protein precursor forms (P2 and P3) may be regulated to follow alternative, mutually exclusive, pathways to generate different sub-sets of biochemical functions from the same type of precursor molecule.
The genera within this family show differences in their genome organisations, particularly in relation to their proteolytic activities. Picornavirus polyproteins contain three ‘recognisable’ types of proteinase - the L, 2A and 3C proteinases (Lpro, 2Apro and 3Cpro, respectively). Additional cleavages (1A/1B and aphtho-/cardiovirus 2A/2B) are mediated by a novel ribosome'skipping' activity : a translational, rather than proteolytic, effect.
Reveiwed by Flint and Ryan (1997) (Download This Paper )